The blog has moved!!!
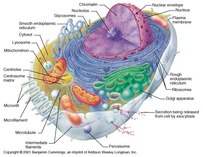
Unit 2 - Cell Structure and Function
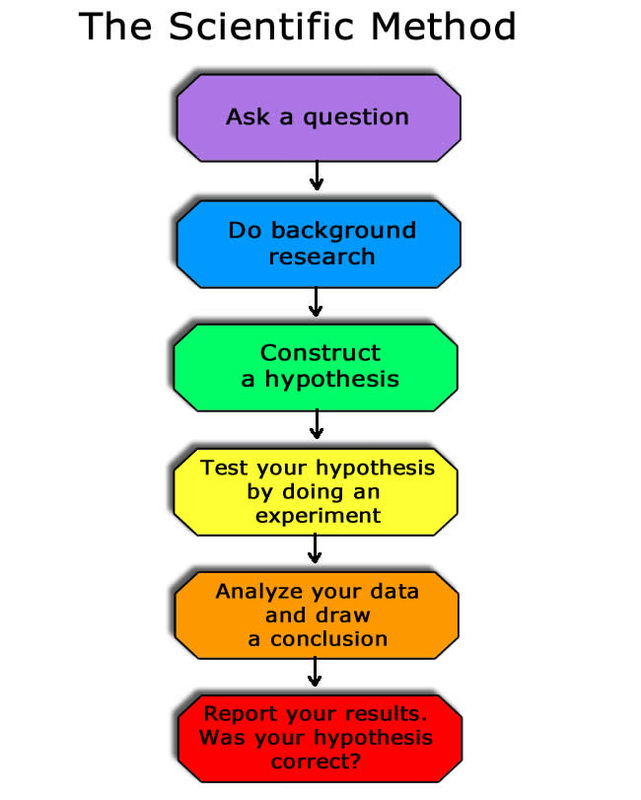
Honors Research Project:
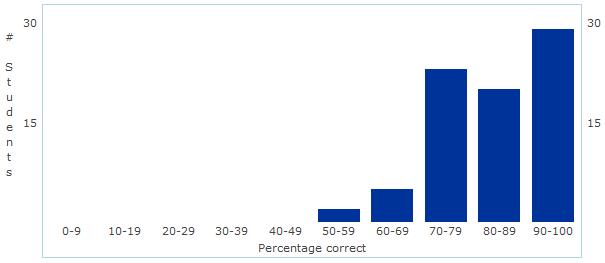
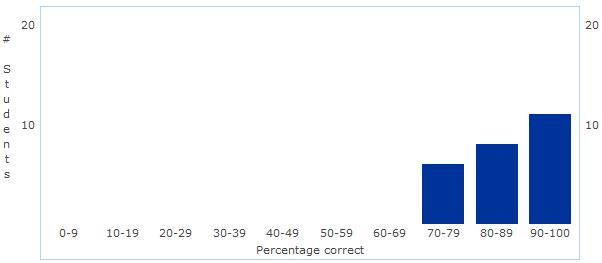
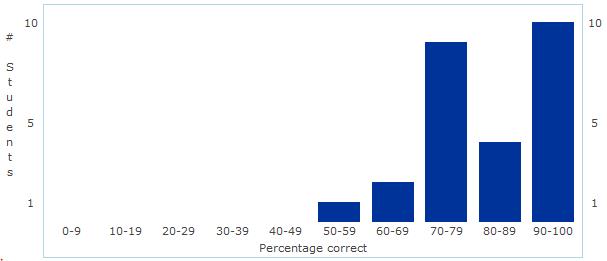
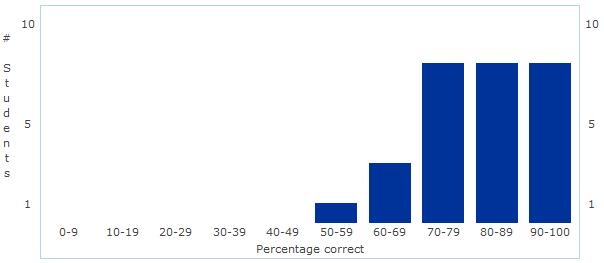
Unit 1 - Chemistry of Life Assessment Statistics Today we discussed the results of our first assessment - Chemistry of Life! Overall, I was fairly pleased with the results. The overall mean and median were 84% - great job everyone!
As we discussed today, I certainly think there is some room for improvement in the manner in which we review and prepare for the assessment - but that will come with time! Good job and let's begin some real "bio" next unit - Cell Structure and Function! Unit 2: Cell Structure and Function Learning Targets (Click link to open) Honors Research Project Work Day! Please fill in the form below with information from the article you selected during class... First Unit Assessment tomorrow!
Make sure you take advantage of the review quiz on Quia! ** Updated 9/7/2014 with Topic Sheet!!! **
Unit 1 - Chemistry of Life Presentation Slides (click to enjoy!)
Organic Macromolecules Key Points:
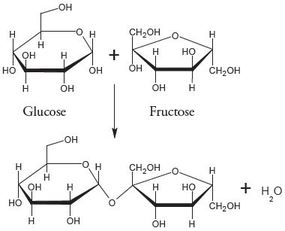
Carbohydrates (sugars) are rings of carbon providing a high amount of short term energy. Think about that candy bar, soda, or even bowl of pasta you just downed - these are high in carbohydrates and will provide you with a short burst of additional energy availability due to our ability to quickly and easily break it down into smaller, usable compounds!
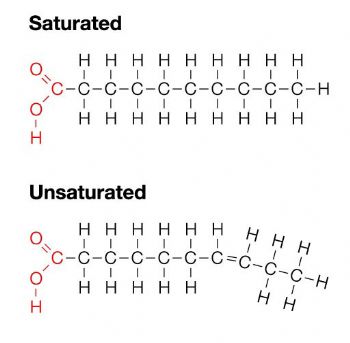
Lipids (fats) are made of long hydrocarbon (hydrogen and carbon) tails extending from a central backbone of hydrogen, carbon, and oxygen. These compounds are hydrophobic (water repelling) due to their lack of overall charge.
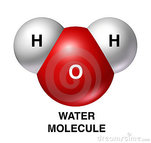
Proteins (amino acids) are made of two consistent groups and one variable "R" side - called a functional group. The Carboxylic Acid Group and Amine Group (see above figure) will be present in every single amino acid. The manner in which each is differentiated into its own, unique amino acid is in the atoms bonded together within the variable "R" functional group. Individual amino acids are also referred to as peptides. These will join together (peptide + peptide + peptide + etc) to form long polypeptide chains called proteins! Nucleic Acids are repeating units that ultimately make up your entire genetic sequence! Once bonded together in long chains - we can call it DNA or RNA depending on its structural organization. We will have an entire unit designated for studying this unique macromolecule as well as its very important roles in maintaining life. Review your Properties of Water:
Purposeful Learning...
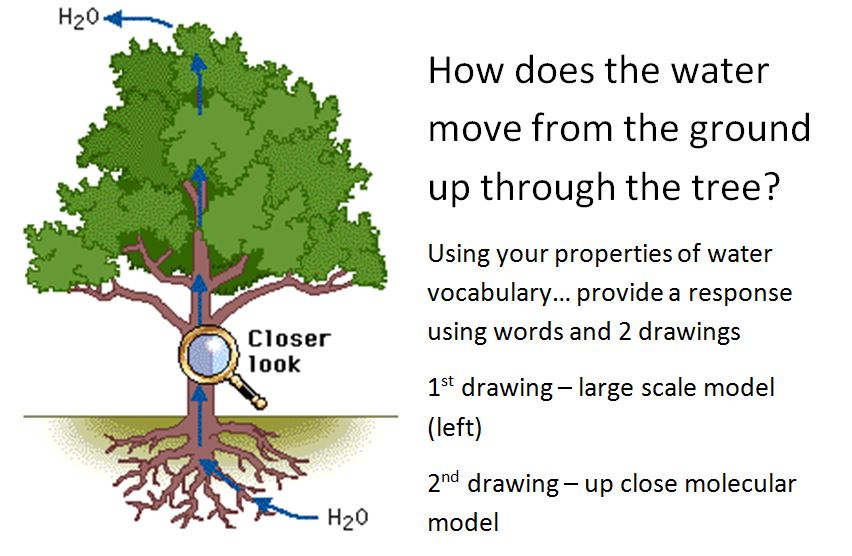
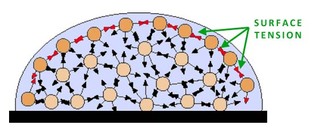
Properties of Water
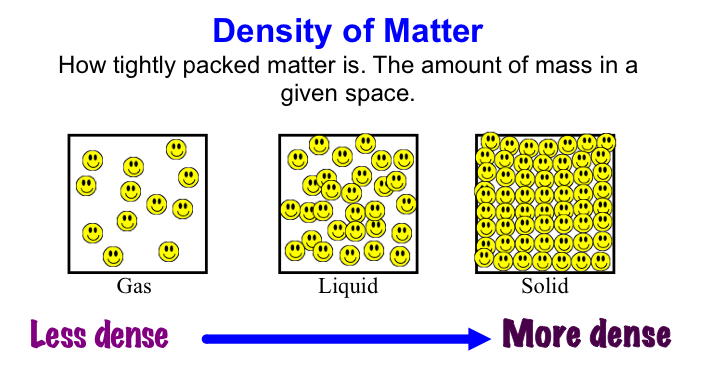
Basilisk Lizard Runs on Water Density: this is a scientific measurement accounting for the number of molecules packed into a set amount of volume. Generally speaking, most gasses are low in density while solids are higher in density. The density of a particular object generally leads to very predictable results. Sometimes, however, density differences can result in some interesting phenomena. First - why does ice float? Shouldn't the solid state of water be more dense and, thus, sink in a glass of water? The air trapped in tap water forces the ice to expand and make the solid form of water less dense! Second - ever heard of underwater lakes and rivers? Wait... that doesn't make sense - underwater lakes and rivers?! Due to different chemical properties, large bodies of liquid (not water!) can form at the bottom of the sea. The underwater "lakes and rivers" are actually large bodies of extremely salty water mixed with hydrogen sulfide. Due to its density (much higher than water), it sinks and forms a new layer! Check the videos below for more details!
To do for Friday - 8/29:
Principal Bacon joined the BVHS Cross Country coaches to complete the ALS Ice Bucket Challenge! Data is your friend!
Dream Big - Work Hard - Stay Humble
Write your story... change history! Ted-Ed Talk by Brad Meltzer Everything you will need to succeed on your final...
Dream - Motivational Video |
Mr. Hulse
Check this blog to recap our daily class topics and activities! Archives
October 2014
Categories
All
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||




















 RSS Feed
RSS Feed
